The density of atom bonds in the different diamond planes defines hardness and cleavage behavior (Fig. 2.15) [BRUN62, FIEL79]. The octahedral plane (111) is the main cleavage plane [FIEL81, LENZ86]. This can be explained by the lower toughness and, therefore, smaller necessary breakage energy along this plane
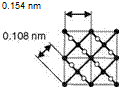 |
Cubic plane (100) Rhombic dodecahedral
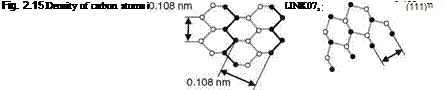
plane (110)
[TELL00, WILK62]. However, cleavage occurs also along the planes (211), (110), and (322) [FIEL79]. Similar mechanisms decide on the lapping behavior of the diamond grits. The smaller the bonding density in a crystal plane is, the easier the material removal. The (110) plane can be machined best via lapping, followed by the (100) plane and (111) as most difficult to machine [BOUW99, YUAZ03].
